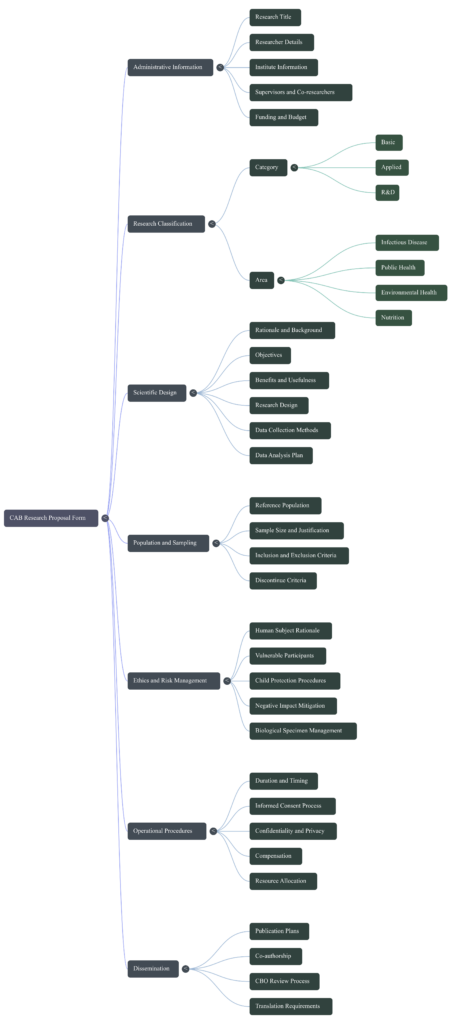
The provided document outlines the mandatory protocols and ethical requirements for investigators conducting research involving human participants through the Community Ethics Advisory Board (CEAB). This governing body, composed of local community leaders and service providers, ensures that all academic inquiries respect the cultural context and protect vulnerable populations within their jurisdiction. Researchers must secure formal approval before beginning their work and are required to provide consistent progress updates and final results to the board. The guidelines emphasize community engagement, requiring that all participant-facing materials be translated into local languages for clarity and transparency. Furthermore, the source details a comprehensive submission package that includes legal permissions, research tools, and credentials to verify the study’s integrity. Ultimately, these rules prioritize local collaboration and ethical accountability to ensure that research benefits and protects the community involved.
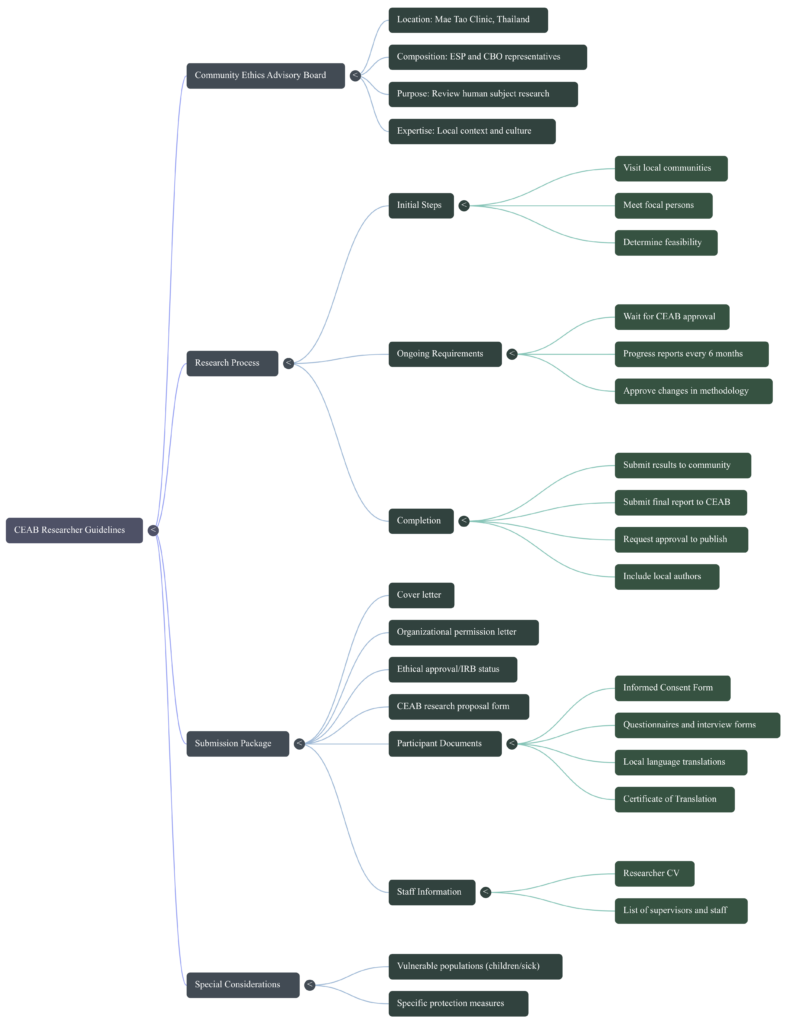
This document is a formal application template used by the Community Ethics Advisory Board at the Mae Tao Clinic to evaluate proposed scientific studies. It requires investigators to provide comprehensive identification details, including their institutional affiliations, funding sources, and specific research categories. The form places a heavy emphasis on ethical safeguards, asking for detailed plans regarding informed consent, the protection of vulnerable participants, and the mitigation of potential risks. Additionally, researchers must outline their methodology, data collection tools, and strategies for maintaining participant confidentiality. The application also tracks logistical needs, such as translation requirements for local languages and future publication intentions. Ultimately, this document serves as a compliance agreement to ensure all projects align with established community ethical guidelines.